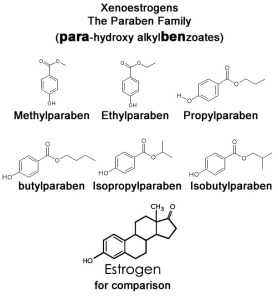
Here we are, back again to the parabens. It may seem like this is a never-ending debate. But that is fine with me. Further debate leads to further investigation. And further investigation may eventually lead us to some solid answers. I’m a scientist. I like solid answers, but truth is ever evasive. As long as everyone is different, there may never be a final answer.
So let’s take a look at the latest information. It comes to use from the Scientific Committee on Consumer Safety (SCCS) through the European Commission. It is an independent non-food scientific committee with no financial gain from industry for their work.
The “Opinion on Parabens” paper can be found here: http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_041.pdf
It is some 35 pages long and gets deep into scientific lingo. So I will try to sort it out for you.
Most parabens are OK. Methylparaben and Ethylparaben are restricted to 0.40% by weight while Propylparaben and Butylparaben are restricted to 0.19% by weight. Oh, if more than one paraben is used, they are restricted to .80% by weight in a formula. Sound good? Most cosmetic/personal care/personal lubricant formulations do not ever use them at that high of a level. The most I have ever seen is around 0.40% by weight when using three of the four.
So looks like we are home and dry? No. We haven’t really even reached for a warm fluffy towel yet.
The Commission wants more data. The rat and mouse models (yes, animal testing) do not have enough correlation to a human model. They are asking for more in vivo human tests. (In vivo means testing on a live creature, in vitro is testing in a petri dish.) There are a few other parabens which have not been tested, but no one uses those in and personal care application.
It appears that parabens whether applied subcutaneously or orally rapidly metabolize into PHBA which is found in all plants and is expected to naturally occur in humans. It is quickly passed through the system through our urine and faeces. However, intact parabens have been found in the urine and/or serum and seminal plasma.
That is why the SCCS set the above stated limits, because they need more data. They set the threshold much higher than the Industry’s recommendation. Enough animal tests, we need to test on humans. Pretty scary, huh? Want to be a human test subject? I would!
Bottom Line Straight from the Report:
With respect to the safe use of parabens as cosmetic ingredients, concern was expressed as to the potential endocrine modifying effects of parabens of higher chain length including Propylparaben, Butylparaben and related iso compounds. Benzylparaben was also of concern. Based upon the currently available in vitro data and in vivo rodent test results, the SCCS agrees that the estrogenic properties displayed by parabens appear to increase with increasing chain length. Nevertheless, the SCCS stresses that the displayed potency levels remain about 3 to 6 orders of magnitude lower than the potency of the positive controls.
So the jury is still out, but the deliberation is drawing near a close. We should have more definitive answers in 4-5 more years. Stay tuned…
Questions? Love Letters? Topics? Nude Pictures? Send them to me at: drtim@quantumcogitation.com